What Is Hydrolyzed polymaleic anhydride (HPMA)
Hydrolyzed polymaleic anhydride is a phosphorus-free water treatment chemical. It forms a chelate with calcium and magnesium ions in water. And hydrolyzed polymaleic anhydride can cause lattice distortion. After the anion is adsorbed by the calcium carbonate nucleus, the nucleus can be charged.
Due to the homophobic electrical repelling, tiny grains are dispersed in the aqueous solution. The precipitate is converted into a loose slag with good fluidity. This phenomenon has a good effect on peeling off the old scale.
Hydrolyzed polymaleic anhydride (HPMA) is compatible with other low-phosphorus corrosion and scale inhibitors such as ATMP and PBTCA. The formed composite corrosion and scale inhibitor can be widely used in the treatment of circulating cooling water, boiler water, oil field water injection, industrial water.
Hydrolyzed Polymaleic Anhydride Structural Analysis
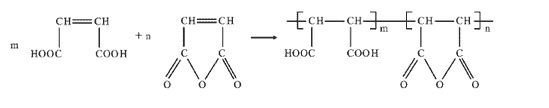
Hydrolyzed polymaleic anhydride (HPMA) is the product of partial hydrolysis of polymaleic anhydride. It has been determined that about 60% to 70% of the polymaleic anhydride is hydrolyzed to a carboxylic acid. That is not completely hydrolyzed.
HPMA is shown by the study of the structure-effect relationship of molecular microstructure. The increase in the degree of hydrolysis is advantageous for HPMA chemical improvement in scale inhibition performance. Therefore, a large degree of hydrolysis is ensured as much as possible during synthesis of hydrolyzed polymaleic anhydride.
From hydrolyzed polymaleic anhydride structural analysis, its carboxyl group in the molecule can form compounds with calcium and magnesium ions in water. And HPMA can promote lattice distortion and turn the precipitation into a loose slag with good fluidity.
Hydrolysed polymaleic anhydride is the same as a polymer having a hydroxyl group or a sulfonic acid group. It can be dissociated into hydrogen ions and acid anions after being dissolved in water. The chain molecules are then negatively charged.
This long-chain negatively charged anion is adsorbed by calcium carbonate nucleus and fine crystal grains. Then bring HPMA with a more negative charge.
The crystallites are dispersed into the aqueous solution due to the homophobic electrical repelling. This prevents the fine grains from coagulating to form larger crystals. Thus, HPMA has a dissolution limiting effect. It has high scale inhibition performance when used at low doses.
In addition, the carbonyl group of HPMA has a strong complexation as an electron donor. This changes the normal morphology of calcium carbonate and calcium phosphate crystals. And can prevent hydrolyzed polymaleic anhydride from growing into a larger crystal. Thereby it has the effect of inhibiting scale formation and peeling off the old scale.
Hydrolyzed Polymaleic Anhydride Characteristics
HPMA chemical is a class of polycarboxylic acid scale inhibitors. It can be matched with other corrosion inhibitors and scale inhibitors. The molecular weight of HPMA is about 600~1200. The aqueous solution is brownish yellow or brownish red, viscous, acidic liquid. Hydrolyzed polymaleic anhydride (HPMA) is chemically stable. It can be used at temperatures below 350 °C.
Hydrolysed polymaleic anhydride has excellent scale inhibition performance for carbonate and phosphate. It has significant solubility effects and significant dispersion effects. HPMA can be adapted to water quality at different pH values. HPMA chemical is phosphorus-free, non-toxic and environmentally friendly. It can be degraded by microorganisms in the natural environment. The degradation products have no effect on the ecological environment.
HPMA Scale Inhibition Performance
The hydrolyzed polymaleic anhydride has the following scale inhibition characteristics.
(1) It is quite effective for calcium carbonate scale. At the same time, it has both lattice distortion and threshold effect (ie, unidirectional conductivity). Even if it is scaled, it is relatively loose. It is easily washed away by water and not formed into a hard scale.
(2) It has high-temperature resistance. Decomposition usually occurs above 350 °C. Can be used at high temperatures.
(3) It has a certain ability to disperse calcium carbonate, calcium phosphate, calcium sulfate and the like.
(4) It can be compounded with a zinc salt. There is a synergistic effect on corrosion inhibition of carbon steel.
(5) It is non-toxic. No significant impact on the environment.
The HPMA chemical corrosion inhibition mechanism is as follows.
The oxygen atom in the polar group (—COOH) and (—OH) in the HPMA molecule can become the adsorption center. It can form a chelate with metal ions in water. Adsorbed on the metal surface. A protective film is formed along the metal surface. Thereby playing a role in corrosion inhibition.
A single polymaleic anhydride is an excellent scale inhibitor. The corrosion inhibition performance is poor. Compared with polyacrylic acid and polymethacrylic acid, HPMA corrosion inhibition performance is slightly stronger. C-P bonds and phosphinic acid groups (-PO2H2) are usually introduced to enhance corrosion inhibition performance.
Compound Scale Inhibitor
1.HPMA-ATMP Compound
ATMP plays a major role as a water quality stabilizer. One is the cooperation of ATMP. It can both soften water and descale. Second, ATMP interferes with the growth of the CaCO3 scale layer. ATMP causes a large distortion of the CaCO3 crystal structure.
2.HPMA-PESA Compound
PESA has a phosphorus-free, non-nitrogen structure. It can be biodegraded in the natural environment. PESA has the dual functions of scale inhibition and corrosion inhibition.
The complex paired calcium carbonate scale of HPMA-PESA has good scale inhibition performance. The concentration, temperature and concentration time of the scale inhibitor have a great influence on the scale inhibition effect.
Increasing the amount of HPMA-PESA compounding agent can also increase the scale inhibition performance of calcium carbonate. When HPMA-PESA is added to a certain amount, it will tend to be saturated and gentle. This composite formulation was the largest at a concentration time of 10 hours. As the concentration-time is further extended, the scale inhibition rate begins to decrease slowly.
3.HPMA-EDTMPS Compound
EDTMPS water miscible energy is chelated with multiple metal ions. A plurality of monomeric structure macromolecular network complexes is formed. EDTMPS destroys the normal crystallization of calcium scale. HPMA-EDTMPS compound scaling rate is greatly improved.
4.HPMA-PAA Compound
PAA is an excellent scale inhibitor and dispersant. It has a good effect of inhibiting the formation of scale and peeling off the old scale. The reaction can be carried out under alkaline and medium concentration multiple conditions without fouling.
As a new type of composite scale inhibitor, HPMA-PAA has the advantages of good scale inhibition effect and good thermal stability.
5.HPMA and HEDP, AMPS Ternary Compounding
The polymer corrosion and scale inhibitor molecules contain two important groups of “stabilizing groups” and “docking groups”. The docking group is adsorbed on the crystallites. Poor water solubility and strong adsorption. The stabilizing group has a good affinity with the solvent.
A large number of carboxyl groups on HPMA are negatively charged after ionization. It is an excellent stable group. The amide group on 2-acryloyl-methylpropane sulfonic acid (AMPS) has strong adsorption to the scale-forming crystallites. It is an excellent docking group. HEDP easily forms a six-membered ring chelate with metal ions and has a synergistic effect.
HPMA is compounded with APMS and HEDP. Excellent docking groups are introduced in the polymaleic anhydride molecule. The adsorption of the polymer is improved. Thereby increasing the “lattice distortion” and “adsorption dispersion” of the polymer. And temperature resistance and timeliness. Very few scale inhibitors can achieve higher scale inhibition rate.
Hydrolyzed Polymaleic Anhydride Synthesis
A 250 mL four-necked flask was charged with a thermometer, a stirrer, a condenser, and a dropping funnel, and a certain amount of maleic anhydride and water was added. The water is warmed to 55 to 60 °C.
After all the maleic anhydride has been dissolved, an appropriate amount of the catalyst is added. The stirrer was turned on while raising the temperature to the reaction temperature. The initiator was added dropwise through a dropping funnel over a period of time.
Pay attention to controlling the temperature during the addition of the initiator. After the completion of the dropwise addition, the reaction was continued for 1 hour to obtain a polymaleic acid product.
TITLE: